Current Research Projects in the Laboratory of Dr. Hallenbeck
Microbial Technologies in
Advanced Biofuels Development
 Advanced biofuels production is necessary to meet the climate change
challenge and the need to secure a future energy supply. My laboratory is carrying out research
projects in a number of areas in this field.
Advanced biofuels production is necessary to meet the climate change
challenge and the need to secure a future energy supply. My laboratory is carrying out research
projects in a number of areas in this field.
Biological
hydrogen production
Hydrogen is poised to
become an important future energy carrier.
Renewable hydrogen production is key to making
it a truly sustainable replacement for fossil fuels, and for realizing its full
potential in reducing greenhouse gas emissions.
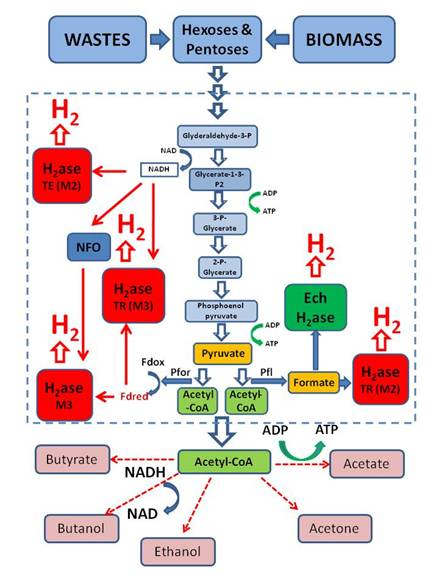
To this end, we have been investigating biological hydrogen 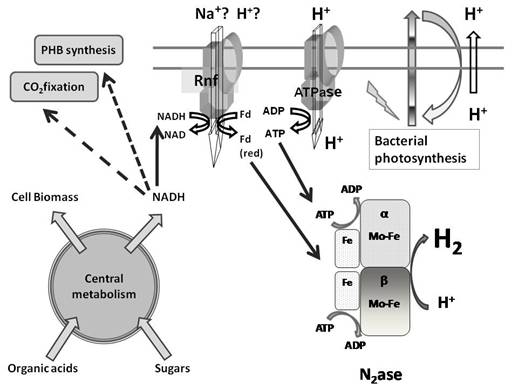 production by microbial fermentation.
This process would use readily available wastes, thus solving another
environmental problem, as well as presently unutilized bioresources,
including enormous supplies of agricultural and forestry wastes. In addition, only relatively simple fermenter
and bioproccess technology is required. In order to make this process practical,
obtainable yields must be increased. We
are carrying out metabolic engineering to create novel strains with increased
yields. We are also developing photofermentation processes, which use light capture by
photosynthetic bacteria, to convert waste glycerol derived from biodiesel, or
sugars derived from cellulose, into hydrogen.
production by microbial fermentation.
This process would use readily available wastes, thus solving another
environmental problem, as well as presently unutilized bioresources,
including enormous supplies of agricultural and forestry wastes. In addition, only relatively simple fermenter
and bioproccess technology is required. In order to make this process practical,
obtainable yields must be increased. We
are carrying out metabolic engineering to create novel strains with increased
yields. We are also developing photofermentation processes, which use light capture by
photosynthetic bacteria, to convert waste glycerol derived from biodiesel, or
sugars derived from cellulose, into hydrogen.
Biofuels
from Algae
We are collecting, isolating
and characterizing algae from different locations in Quebec for possible use in
biofuels production. In particular, we
are looking for algae naturally high in lipids which could be converted to biodiesel. Eventually it might be possible to produce
biofuels in a renewable manner by situating algal ponds near industrial
facilities producing waste heat and carbon dioxide which could be fixed and
converted to a
biofuel. Using a waste stream as water
source will reduce the cost of nutrients for the algal culture, reduce the
demand on clean water supplies, and provide waste treatment.
Regulation of Nitrogen
Fixation in the Photosynthetic Bacteria
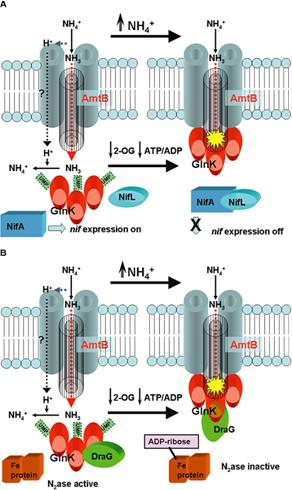 Nitrogenase in the
photosynthetic bacteria is tightly controlled at three levels. We are investigating how the bacterium senses
changes in its environment and regulates nitrogenase through covalent
modification and metabolic flux. At
present, we are concentrating on the mechanism of action of AmtB, a membrane
bound sensor which is a member of a family of proteins widely found in
bacteria, archaea , and eucaryotes. In the photosynthetic bacteria, AmtB appears
to function by changing its interaction with PII proteins, soluble cytoplasmic
proteins that play key roles in the regulation of nitrogen metabolism. We are targeting key amino acids in AmtB by
site-directed mutagenesis and studying the effects of the introduced changes on
ammonium transport, interaction with PII proteins, and “switch-off” of
nitrogenase.
Nitrogenase in the
photosynthetic bacteria is tightly controlled at three levels. We are investigating how the bacterium senses
changes in its environment and regulates nitrogenase through covalent
modification and metabolic flux. At
present, we are concentrating on the mechanism of action of AmtB, a membrane
bound sensor which is a member of a family of proteins widely found in
bacteria, archaea , and eucaryotes. In the photosynthetic bacteria, AmtB appears
to function by changing its interaction with PII proteins, soluble cytoplasmic
proteins that play key roles in the regulation of nitrogen metabolism. We are targeting key amino acids in AmtB by
site-directed mutagenesis and studying the effects of the introduced changes on
ammonium transport, interaction with PII proteins, and “switch-off” of
nitrogenase.